1. Introduction
Acacia rigudula
Benth., blackbrush, is a shrub found growing on rocky ridges in
southwest and west Texas, and in the northern states of Mexico,
including Tamaulipas, Nuevo Leon, Chihuahua, San Luis Potosi, and
Jalisco [1]. The leaves are
bipinnate and predominately short 15–25 mm long, pinnae 1–2 pairs that
are 6–15 mm long, with 2–4 pairs (rarely 5 pairs) of elliptic to oblong
leaflets 4–12 mm long and 6–8 mm wide. Blackbrush is heavily defended
with long sharp spines and, if left unchecked, will form virtually
impenetrable thickets. In addition to its physical defences, A. rigidula has been shown to contain appreciable levels of toxic alkaloids. Sheep and goats grazing on a related species, Acacia berlandieri
Benth., guajillo, during periods of drought in the Rio Grande Plains of
Texas have developed a locomotor ataxia referred to as guajillo wobbles
or limber leg [2]. Previous analysis of blackbrush had detected and identified four amines, N–methyl-β-phenethylamine (NMPEA), tyramine, N-methyltyramine, and hordenine, which were also found in guajillo [3].
The locomotor ataxia developed in the early A. berlandieri feeding trials [4] was not observed in adequately fed animals injected with NMPEA or tyramine [5]. A review [6]
of previous work has shown that many plant species endemic to southwest
Texas and northern Mexico contain a wide variety of aromatic
monoamines. A need to more fully explain the results of earlier studies,
together with advances in GC-MS technology, led to an intensive
chemical analysis of A. berlandieri to identify other amines and alkaloids present in the leaves [7].
As a result of this study, an intensive chemical analysis of A. rigidula Benth. was undertaken to identify the amines and alkaloids present in the leaves and stems that would comprise browse material.
2. Results and discussion
Leaves and stems that would comprise browse material were packed into an
extraction thimble, placed in a Soxhlet apparatus and extracted with
methanol followed by extraction with chloroform.
This extraction procedure was compared with the more traditional
methods of extraction by soaking the plant material in acid solution [2, 8]
and found to produce a cleaner, more complete isolation of the amines
and alkaloids present in the sample. The initial extract was
fractionated by extraction with aq. acid, the pH of the acid extract was
adjusted to ca pH 10 prior to re-isolation of the alkaloids by
extraction with organic solvents. The pH of the aq. fraction was
routinely checked between extractions and adjusted (dil. NaOH added) as
necessary. It was found that when the pH exceeded 11, a large portion of
the phenol containing alkaloids remained in the aqueous solution as
their corresponding phenolate salts. Particular care was taken to
maintain the extracts and isolates under an inert atmosphere. Left
unprotected, the isolated amines and alkaloids readily decomposed.
Forty-four amines and alkaloids, including the four previously encountered amines, N-methyl-β-phenethylamine, tyramine, N-methyltyramine,
and hordenine, were identified by GC-MS. Both splitless injection and
dedicated on-column injection systems were employed for GC-MS analysis.
The on-column injection is a much milder method of sample introduction.
This technique, although it produced the most complex chromatograms, was
best for the detection of the phenol containing components of the
mixture which tended to be more thermally labile.
No derivatization was performed on the analytes, therefore GC-MS
analysis would only be expected to detect the volatile amines and
alkaloids present in the sample.
The majority of the isolated alkaloids, 29 of the 44 identified, were related to the parent compound β-phenethylamine. These compounds generally varied in the degree of N-methylation, 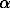 -methylation (amphetamine family), and in oxygenation of the aromatic ring (tyramine, dopamine, and mescaline families).
The 2-cyclohexylethylamine and the N-cyclohexylethyl-N-methylamine are the saturated analogs of the phenethylamine and NMPEA respectively. Tryptamine, N-methyltryptamine, and N,N-dimethyltryptamine were also isolated from blackbrush. Tryptamine and N,N-dimethyltryptamine were also detected in the related species guajillo [7], albeit at relatively low levels. Early season levels of tryptamine in A. berlandieri range from 90 to 124 ppb while late season levels range between 287 and 334 ppb. N,N-Dimethyltryptamine
was only detected in late season guajillo at levels ranging between 75
and 115 ppb. It is interesting to note that a similar animal ataxia
called Phalaris staggers occurs in Australia and is associated with
consumption of various Phalaris species which are known to contain N,N-dimethyltryptamine [9].
Other noteworthy alkaloids found in blackbrush include nicotine,
nornicotine, and four tetrahydroisoquinoline alkaloids, anhalamine,
anhalidine, anhalonidine, and peyophorine.
The amides of the amino acids pipecolic acid and p-hydroxypipecolic
acid were also detected. It should be noted that no other amino acid
esters or amides were detected in the extract.
-methylation (amphetamine family), and in oxygenation of the aromatic ring (tyramine, dopamine, and mescaline families).
The 2-cyclohexylethylamine and the N-cyclohexylethyl-N-methylamine are the saturated analogs of the phenethylamine and NMPEA respectively. Tryptamine, N-methyltryptamine, and N,N-dimethyltryptamine were also isolated from blackbrush. Tryptamine and N,N-dimethyltryptamine were also detected in the related species guajillo [7], albeit at relatively low levels. Early season levels of tryptamine in A. berlandieri range from 90 to 124 ppb while late season levels range between 287 and 334 ppb. N,N-Dimethyltryptamine
was only detected in late season guajillo at levels ranging between 75
and 115 ppb. It is interesting to note that a similar animal ataxia
called Phalaris staggers occurs in Australia and is associated with
consumption of various Phalaris species which are known to contain N,N-dimethyltryptamine [9].
Other noteworthy alkaloids found in blackbrush include nicotine,
nornicotine, and four tetrahydroisoquinoline alkaloids, anhalamine,
anhalidine, anhalonidine, and peyophorine.
The amides of the amino acids pipecolic acid and p-hydroxypipecolic
acid were also detected. It should be noted that no other amino acid
esters or amides were detected in the extract.
Initial
identification of the alkaloids present within the plant extracts was
based upon library comparison of their MS fragmentation patterns with
the final confirmation of identification made by direct comparison with
spectra of authentic samples. Quantification of the levels of each
compound was based upon standard curves generated with authentic
samples. Leaves, petioles and unlignified stems were collected twice, a
first growth sample collected early in the spring and a late season
sample collected before frost in the autumn. As was previously found
with NMPEA the foliage collected in the autumn contained higher
quantities of amines and alkaloids [10]. There was also a distinct increase in the number and quantity of methylated analogs present (Table 1).

Table 1. Amines and alkaloids from A. ridigula Benth.


Several as yet unidentified amine-containing compounds have been
detected but have not yet been identified. It is also probable that
several biosynthetic precursors are present in the mixture but as yet
are unresolved and are below the threshold of GC-MS detection.
Phenolic amines, as a group, impact the hypothalamic–pituitary–adrenal axis [11]. The consequent release of ACTH and cortisol results in symphathomimetic action. The number of phenolic amines reported in Table 1
and their concentrations in the plant indicate a substantial toxic load
to animals consuming blackbrush. The toxicity of nicotine and
nornicotine has been well established [12],
as has the psychoactivity of mescaline and its derivatives. None of the
compounds identified appear to have been implicated in locomotor
ataxia. However the presence of the amphetamines suggests the
possibility for a reduction of monoamine oxidase activity [13].
3. Experimental
GC-quadrupole
MS (EIMS at 70 eV) were obtained on Hewlett Packard Model 5988A and
Model 5970C systems each having both splitless heated injection port and
dedicated on-column injection capabilities. Grade 0 He was used as the
carrier gas and the transfer line was maintained at 280°. Heated
injection conditions include: head pressure 5 psi; injection volume 1 μl; injection port 200°; splitless time 1 min; purge flow 60 ml/min; initial time 1 min; ramp 3°/min; final temp. 270°; final time 20 min. On-column injection conditions include: head pressure 5 psi; injection volume 1 μl; initial temp. 60°; initial time 1 min; ramp 1.5°/min;
final temp. 270°; final time 35 min. WCOT cross-linked methyl silicone
12 (only used on HP 5970 system) and 36 m, 0.2 mm i.d., 33 μ
coating in thickness capillary columns were used. Mass spectral data
was collected as the total ion chromatograms in the operating range of
35–800 amu. The GC-MS operating system including the Wiley mass spectral
library and the NBS mass spectral library of standards. Preliminary
identification was made by library comparison, final identification was
made by direct spectral comparison with the spectra of an authentic
sample obtained from the GC-MS. Authentic samples were either purchased
or prepared by known chemical procedures.
GC-MS quantification was made by using the area under a selected m/z
peak for each compound and comparing this to a standard curve generated
by injection of a series of known standards. Care was taken to make
sure that the detector response was linear in the concentration range
being run.
3.1. Collection of leaves and stems
Samples of A. rigidula
Benth. were collected from plants growing on a southwest-facing slope
in Zavala County, Texas. Early season collection was performed in the
spring after vigorous new growth appeared. Late season collection was
performed in the late fall prior to the first front and before color
change was detected. Leaves, petioles, and attached tender stems were
collected until ca 500 g of fr. wt had been gathered. The material was
sealed in waterproof bags and placed on ice immediately. Samples were
then frozen at -20° and stored until extraction. Voucher specimens were
collected and stored at the Texas A and M University Agricultural
Research and Extension Center, Uvalde, Texas.
3.2. Soxhlet extraction procedure
Still frozen A. rigidula was packed into Whatman single thickness cellulose extraction thimbles (ca 50 g/thimble, 200 g total) and extracted continuously for 24 h with MeOH. The MeOH was removed and replaced with CHCl3
and the extraction continued for an additional 24 h. A pilot study
established that the extracts could be safely concentrated by rotary
evaporation (water aspirator). The MeOH extract was concentrated and the
residue dissolved in 100 ml of CHCl3. This solution was
extracted ×3 with 50 ml portions of 10% aq. HCl. The acid fractions were
combined, the pH adjusted to ca 10.3 by addition of aq. NaOH, and the
resulting solution was first extracted ×3 with 50 ml portions of CHCl3 followed by extraction ×3 with 50 ml portions of EtOAc. These organic extracts were combined, dried with MgSO4, filtered, concentrated under vacuum, and stored under argon prior to analysis by GC-MS. The CHCl3
fraction from the Soxhlet extraction was handled in an identical
fashion to the MeOH extract with the exception that it was not first
concentrated prior to aq. acid extraction. The MeOH and CHCl3 extracts from the Soxhlet extraction were not combined.
3.3. Acid extraction of plant material
Acacia rigidula,
100 g, was placed into a 1 l Erlenmeyer flask and mixed with 500 ml of
10% aq. acid (HCl and HOAc were both used). The suspension was stirred
under argon at 60° overnight. The darkened suspension was filtered
through glass wool covered with a 2 cm bed of sand. The filtrate was
extracted ×2 with 100 ml portions of EtOAc followed by ×3 extraction
with 100 ml portions of CHCl3. The pH of the filtrate was
adjusted to ca 10 by addition of NaOH pellets. This solution was
extracted ×3 with 150 ml portions of CHCl3 followed by extraction ×3 with 150 ml portions of EtOAc. The organic extracts were combined, dried over MgSO4, filtered, concentrated under vacuum and stored under argon prior to analysis by GC-MS.

References
1. Vines, R. A., Trees, Shrubs, and Woody Vines of the Southwest. University of Texas Press, Austin, TX, 1984.
2. D. A. Price and W. T. Hardy. Am. Vet. Med. Assoc 26 (1953), p. 222.
3. Adams, H. R. and Camp, B. J., Toxicon, 1966, 4, 85.
4. Broughton, I. B. and Hardy, W. T., 54th Ann. Rep., Texas Agric. Exper. Stat., 1941, p. 159.
5. T. D. A. Forbes, R. D. Randel, D. R. Tolleson and C. M. Hensarling. Afric. J. Anim. Sci 23 (1993), p. 196.
6. Smith, T. A., Phytochemistry, 1977, 16, 9.
7. Clement, B. A., Goff, C. M. and Forbes, T. D. A., Phytochemistry, 1997, (in press).
8. B. J. Camp and J. A. Moore. Am. Pharm. Assoc 49 (1996), p. 158.
9. H. J. Lee and R. E. Kuchel. J. Agric. Res 4 (1953), p. 88.
10. Forbes, T. D. A., Pemberton, I. J., Smith, G. R., Hensarling, C. M. and Tolleson, D. R., Texas Agricultural Experiment Station Consolidated Progress Report No. 4875, 1991.
11. H. R. Vera-Avila, R. D. Randel and T. D. A. Forbes. Anim. Endocrinol. 13 (1996), p. 285.
12. Duke, J. A., Handbook of Medicinal Herbs. CRC Press, Boca Raton, FL.
13. T. J. Mantle, K. F. Tiptons and N. J. Garret. Pharm 25 (1976), p. 2073.

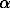 -methylation (amphetamine family), and in oxygenation of the aromatic ring (tyramine, dopamine, and mescaline families).
The 2-cyclohexylethylamine and the N-cyclohexylethyl-N-methylamine are the saturated analogs of the phenethylamine and NMPEA respectively. Tryptamine, N-methyltryptamine, and N,N-dimethyltryptamine were also isolated from blackbrush. Tryptamine and N,N-dimethyltryptamine were also detected in the related species guajillo [
-methylation (amphetamine family), and in oxygenation of the aromatic ring (tyramine, dopamine, and mescaline families).
The 2-cyclohexylethylamine and the N-cyclohexylethyl-N-methylamine are the saturated analogs of the phenethylamine and NMPEA respectively. Tryptamine, N-methyltryptamine, and N,N-dimethyltryptamine were also isolated from blackbrush. Tryptamine and N,N-dimethyltryptamine were also detected in the related species guajillo [